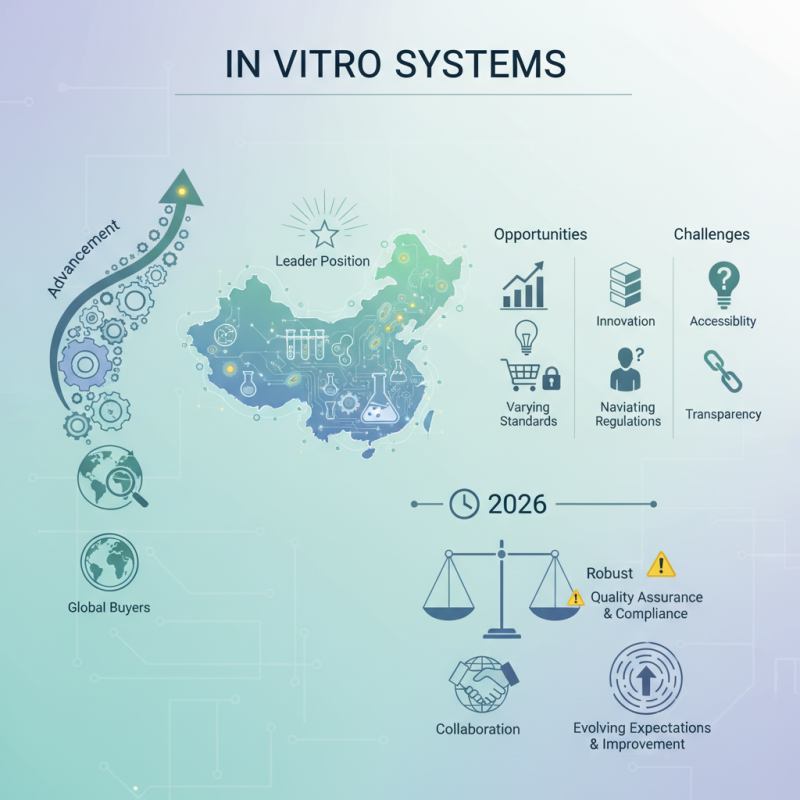
The advancement of the "in vitro system" has transformed the landscape for global buyers. As China positions itself as a leader in this field, the implications for innovation and accessibility are profound. This growing industry boasts cutting-edge technologies, yet challenges remain. Many buyers must navigate varying standards and expectations.
In 2026, the global demand for reliable and efficient in vitro systems will likely intensify. Companies in China are stepping up, offering robust products that cater to diverse needs. However, prospective buyers should approach with caution. Understanding the nuances of quality assurance and regulatory compliance is critical.
Chinese manufacturers have made significant strides, showcasing expertise and reliability. Yet, the learning curve for international buyers persists. As expectations evolve, there is room for improvement in transparency and communication. This dynamic environment offers both opportunities and challenges for those seeking the best in vitro system solutions.
China’s role in the global in vitro diagnostic market is becoming increasingly significant. According to a recent market report, the in vitro diagnostics sector in China is projected to grow at a compound annual growth rate (CAGR) of 9.5% from 2021 to 2026. This growth is driven by advancements in technology, increasing healthcare demands, and supportive government policies. Chinese manufacturers are focusing on enhancing their product quality and innovating their technologies, making them attractive to global buyers.
One of the biggest challenges facing the industry is quality assurance. While many Chinese companies are making strides, some still struggle with meeting international standards. Reports indicate that around 30% of diagnostic products from China were questioned for quality in previous years. Companies must enhance transparency and quality control measures to build trust with international markets.
Tip: When considering procurement, prioritize suppliers who have certifications from reputable organizations. This helps ensure that the products meet global standards.
Additionally, the COVID-19 pandemic has spurred innovation in diagnostics, from rapid testing kits to advanced molecular testing platforms. The market's dynamism highlights the necessity for continuous improvement. Chinese firms need to remain agile and responsive to global demands while working on areas that require further development.
Tip: Stay informed about emerging trends and regulatory changes in the market. This knowledge can provide a competitive edge.

China is emerging as a leader in in vitro system development. Rapid technological advancements are a key factor in this growth. Innovative research institutions are increasing in number and quality. They focus on creating effective and efficient solutions. Collaboration between academic and industrial sectors enhances this potential. A strong emphasis on research and development drives the market forward.
China's regulatory environment is becoming more supportive. It has made strides in improving approval processes. This encourages investment in biotechnology sectors. However, challenges remain. Regulatory requirements can still be complex and time-consuming. Furthermore, there is a need for better standardization practices. This inconsistency can deter potential global buyers. Addressing these issues is crucial for sustained growth and attraction.
The nation's vast talent pool is another significant advantage. Scientists and researchers are increasingly being trained in cutting-edge techniques. However, a disconnect sometimes exists between research findings and practical applications. Bridging this gap is essential for effective commercialization. As these factors continue to evolve, China's in vitro systems may play a pivotal role in the global market.
Chinese manufacturers are making significant strides in in vitro technologies. Innovations are blooming in various sectors, including diagnostics and research tools. These advancements focus on improving accuracy and efficiency. Many products are now more user-friendly and cost-effective. This shift positions China as a key player in the global market.
Research and development efforts highlight the commitment to quality and reliability. New techniques are emerging that enhance cell culture and assay performance. However, some challenges remain. The need for rigorous quality controls is critical. Not all products meet international standards, prompting ongoing evaluation.
Collaborations between Chinese firms and global partners are increasing. This phenomenon allows knowledge sharing and technology transfer. While the potential is immense, there are gaps that require attention. Continuous improvement is essential for maintaining competitiveness. The journey towards excellence in in vitro systems is ongoing.
China offers a unique landscape for global buyers in the in vitro industry. However, challenges abound. Language barriers can hinder effective communication. Navigating regulations requires expertise. Buyers often feel overwhelmed when trying to understand local standards. Thus, thorough research is vital.
Opportunities also exist for those willing to invest the effort. China's robust manufacturing capabilities provide cost advantages. The rapid technological advancements in research and development create potential partnerships. Buyers can tap into these innovations by understanding local needs. Building relationships with suppliers can lead to improved product quality and reliability.
Nonetheless, the differences in market dynamics can be daunting. Buyers must remain adaptable and open-minded. Developing cultural competence is essential to navigate these complexities. Without it, misunderstandings may arise, impacting business deals. Recognizing these factors can facilitate a smoother experience for global buyers in China's in vitro market.
This bar chart illustrates key projections for the In Vitro System market in China for global buyers by 2026, covering estimated market size, expected growth rate, and export potential. These insights can help global buyers navigate opportunities and challenges in the Chinese market.
China's in vitro system is gaining attention as we approach 2026. The country is leveraging advanced technologies to enhance its production capabilities. Innovations in automation and artificial intelligence are transforming the industry. Laboratories are becoming more efficient and precise, leading to better outcomes.
However, challenges persist. Quality control remains a significant concern. As demand increases, ensuring consistency becomes more complicated. The need for regulatory compliance is critical, yet adaptation can be slow. Balancing rapid growth with stringent standards is essential for long-term success.
Looking ahead, partnerships between public and private sectors are vital. Collaboration can drive research and development. This could lead to breakthroughs in various applications, such as diagnostics and therapeutics. Yet, there is still much to explore. Increased focus on sustainability and ethical practices is necessary. Striving for these ideals can help shape a better future for in vitro systems in China.
| Dimension | 2023 | 2024 | 2025 | 2026 |
|---|---|---|---|---|
| Market Size (Billion USD) | 10 | 12 | 15 | 18 |
| Growth Rate (%) | 8 | 10 | 12 | 15 |
| Key Application Areas | Diagnostics, Research | Diagnostics, Drug Development | Diagnostics, Personalized Medicine | Diagnostics, Therapeutics |
| Regulatory Changes | Current Framework | Evaluation Updates | New Guidelines | Harmonization Efforts |
| Key Innovations | Automation | AI Integration | Lab-on-a-Chip | Point-of-Care Testing |